How the Retina Works *[1]
[1] This chapter is an update of a 2003 American Scientist review article by Helga Kolb (1).
Abstract
Much of the interpretation of our visual world takes place in the retina itself through the actions of specialized neural circuits. Rods and cones, whose distributions and interactions with other retinal neurons have evolved differently among species, form the initiating elements of these circuits. Neurons within the retina, horizontal cells, amacrine cells, bipolar cells, and ganglion cells, extract salient elements from the rod and cone sensory layer using patterns of selective excitatory and inhibitory, often novel, synaptic connections within the retinal synaptic layers. The retina segregates these extracted image elements into parallel output pathways specifying image features and transmits them for further analysis to brain visual centers. The system is adaptive to conditions of illumination, sometimes employing locally diffusing neuromodulators to signal global changes in scene. All this image processing occurs within a photosensitive sheet of neural tissue less than half a millimeter thick. This review previews and highlights the content of Webvision, a tutorial website containing a set of chapters written by experts in the field on the elements of visual-system anatomy and function, focusing on retina, its embryonic development, image processing capabilities, disruptions in disease, prospects for cure, and implications for nervous system research.
Introduction
The retina is a filmy piece of tissue, barely half a millimeter thick, that lines the inside of the eyeball. The tissue develops from a pouch of the embryonic forebrain, and is therefore considered part of the brain (see Webvision section ‘Retinal Neurogenesis: Early stages in the Development of Neurons and Pathways’). The retina has a basic structure like that of a three-layer cake, with the bodies of nerve cells arrayed within layers and each layer separated by densely packed ‘frosting’ or ‘plexiform layer’ of synaptic connections (Figure 1). The retina includes both the sensory neurons that respond to light and intricate neural circuits that perform the first stages of image processing. Ultimately, extracted aspects of the image are encoded as electrical messages which travel through the optic nerve and into the brain for further feature identification and visual perception.

Figure 1. Diagram of a human eye shows its various structures (left). A thin piece of retina is enlarged in a photomicrograph (right), revealing its layers. The photoreceptors lie against a dark row of cells called the pigment epithelium. (Drawing by Helga Kolb. Except where noted, photographs by Nicolas Cuenca and Helga Kolb).
Logically, one might expect that the surface of the retina (the layer exposed to the liquid in the eyeball’s vitreous chamber) would be closest to the light and would contain the sensory cells, the photoreceptors, but actually these cells lie at the very back of the retina; light rays must pass through the entire retina before reaching light-sensitive visual pigment molecules to initiate the sensory process. This is because the photopigment-bearing membranes of the photoreceptors must be in contact with the pigment epithelial layer at the back of the eye, which provides a steady stream of the vital molecule retinal, (vitamin A aldehyde). Retinal becomes covalently bound to the photoreceptor opsin proteins, where this small molecule changes its conformation from 11-cis to all trans in response to excitation by photons (2), which are the quantal packets that make up light (3). The cis to trans isomerization of retinal triggers a shape change in opsin molecules (Figure 2, lower right). This initiates a cascade of biochemical reactions within rods and cones that constitute phototransduction, the first steps in vision (see Webvision chapters ‘Photoreceptors’ and ‘Phototransduction in Rods and Cones’). Retinal molecules are then recycled back into the pigment epithelium, where they are re-isomerized from trans to cis conformation (4).
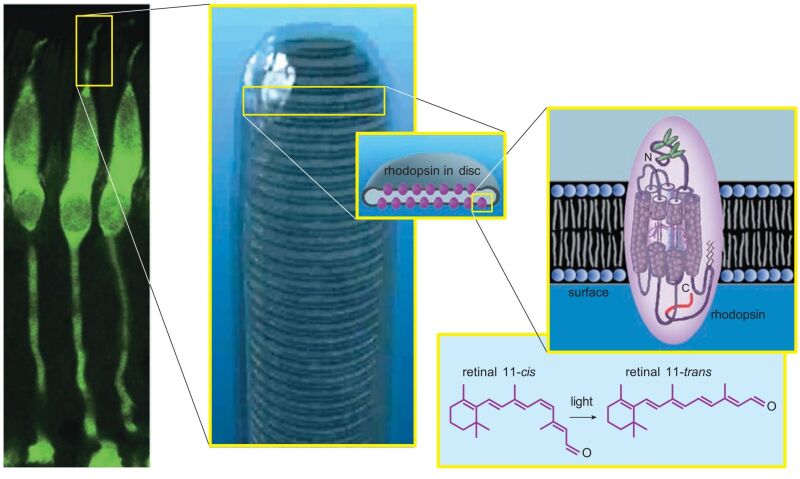
Figure 2. Cone photoreceptors from a monkey are stained with a fluorescent green dye (left). When the outer segments of cones or rods are magnified further, stacked membrane disks are visible inside (middle). The disks are studded with thousands of rhodopsin molecules. Each rhodopsin consists of a membrane-traversing protein with a molecule of retinal covalently bound in its core (right). When exposed to light, the 11-cis bond in the retinal molecule rotates, nudging the opsin protein to also change shape, initiating the process of phototransduction (lower right). (Middle photograph courtesy of Carlos Rozas).
Pigment epithelial tissue behind the retina is usually very dark because these cells are full of melanin granules. The pigment granules absorb stray photons, preventing reflection back into the photoreceptors, which would cause images to blur. They protect the cells from too much exposure to light radiation, and additionally maintain ionic homeostasis within the retina (see Webvision chapter ‘The Retinal Pigment Epithelium’).
Retina Design According to Lifestyle
All vertebrate retinas contain at least two types of photoreceptors—rods and cones. Rods generally serve low-light, gray-scale vision while cones serve daylight and colored vision. Variations among the retinas of different species reveal both adaptations to the different environments in which they live and their evolutionary history (5, 6). Most fish, frog, turtle, and bird retinas have three to five types of cones and consequently very good color vision. Keep in mind that reptiles and fish are “cold blooded” and need to be active in the warmth of daytime. Most mammals have retinas in which rods predominate (see Webvision chapter ‘Photoreceptors’).
When the number of mammals started to explode as the dinosaurs died out, their previous habitat was likely a dark place; perhaps they had been nocturnal. Contributing to dinosaur demise, the Earth may have become cold and possibly covered in ash and clouds from an asteroid strike. The tiny, fur-covered early mammals were already adapted to generate their own body heat and had evolved visual systems sensitive to dim light. Modern rodents such as rats and mice, which are nocturnal animals, still have retinas overwhelmingly dominated by rods; their cones are few and make up only 3 to 5 percent of their photoreceptors (7).
High acuity cone regions
Most mammalian retinas continue to have a preponderance of rods, and the cones are often concentrated in specialized regions. In species such as cats and dogs, images focus to a central specialized area, aptly called the area centralis, where cones attain high density (8). The retinas of mammals such as rabbits (9) and squirrels, as well as those of some nonmammals like turtles, have a long, horizontal strip of specialized cells called a visual streak, which can detect the fast movement of predators along the visual horizon (Figure 3). Rodents show spatial specialization expressing short-wavelength (S- or UV-cones) in ventral retina but middle-wavelength (M-cones) in dorsal retina (10). Primates as well as some birds and mammalian carnivores like the cat have front-projecting eyes allowing binocular vision and thus depth perception. The eyes of primates and most birds are specialized for good daylight vision and can discriminate color and fine details. Primates and raptors, like eagles and hawks, have a forward-facing fovea, a tremendously cone-rich pit in the retina devoid of rods where images focus and fine details of image, color and depth are perceived. Some fish (11) and lizards (12, 13) also have foveas.
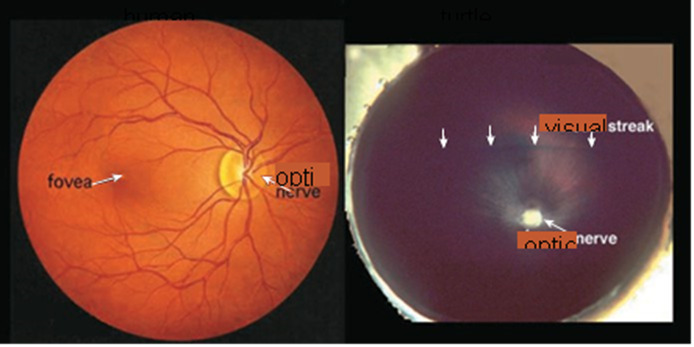
Figure 3. An ophthalmologist’s view of the human retina (left) shows the optic nerve head, from which blood vessels radiate to nourish every part of the tissue, and shows the fovea, which is where images focus most directly and is an area specialized for sharp daylight vision. The fovea is densely packed with more than half of the human cone photoreceptors. The turtle retina (right) has no blood vessels radiating from the optic nerve and no fovea. Instead, it has a visual streak, a region of specialized cells running horizontally above the optic nerve, which can detect fleeting movements and orient those movements relative to the horizon. Regional differentiation is a characteristic of retinas.
Balancing rod and cone vision
Primates, in fact, have what is called a duplex retina, allowing good visual discrimination in all lighting conditions. The fovea contains most of the cones, packed together as tightly as physically possible, and allows good daylight vision (see Webvision chapter ‘The Architecture of the Human Fovea’). More peripheral parts of the retina are packed with rods and can detect the slightest glimmer of photons at night. Most mammals have two types of cones, green-sensitive and blue-sensitive, but primates have three types—red-sensitive as well as the other two (Figure 4). With our primate cone vision, we can see from gray dawn to the dazzling conditions of high noon with the sun burning down on white sand. To accomplish this, as a first step, the cone photoreceptors themselves can adapt to the surrounding brightness, and circuitry through the retina can further extend the eye’s cone response. Similarly, the rod photoreceptors and the neural circuitry to which they connect can adapt to lower and lower intensity of light (14) providing vision even on moonless nights.
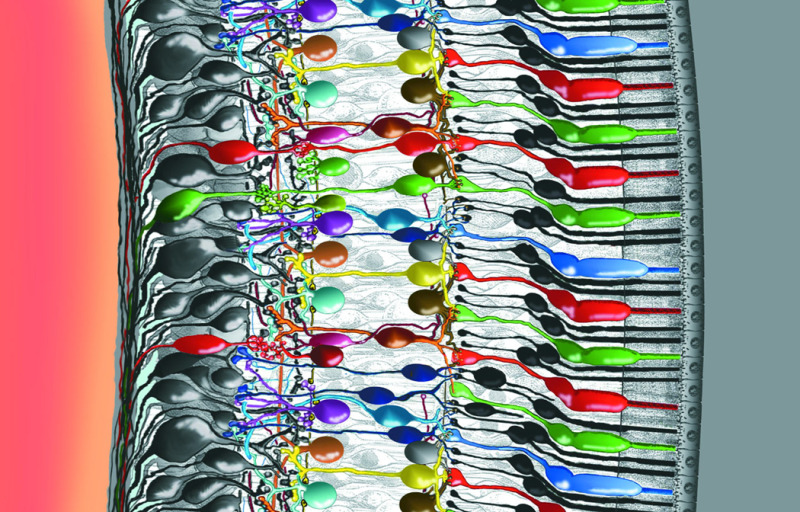
Figure 4. Intricately wired neurons in the retina allow a good deal of image processing to take place in the eye itself. Some of the interactions among the cells in this delicate piece of tissue are well understood, but others not. In this rendering, light enters the eye from the left. The photons travel through the vitreous fluid of the eyeball and penetrate the entire retina, which is less than half a millimeter thick, before reaching the photoreceptors—the cones and rods that absorb photons and respond to light (the colored robust cones and slender black elongate rods attached to the epithelium at right). Signals then pass from the photoreceptors back to the left through a series of neural connections toward the surface of the retina, where the ganglion-cell nerve-fiber layer relays the processed information to the optic nerve and into the brain (Drawing by Helga Kolb).
Anatomy and Physiology
Understanding the anatomy of the retina is essential to understanding its function (Figure 5). The photoreceptors lie in a layer against the back of the eyeball. Multiple types of image-processing retinal interneurons lie in the inner nuclear layer, the middle of three cell layers. In this layer there are one to four types of horizontal cells (see Webvision chapter ‘S-Potentials and Horizontal Cells’), 11 types of bipolar cells (see Webvision chapter ‘Bipolar Cell Pathways in the Vertebrate Retina’) and 22 to 30 types of amacrine cells (see Webvision chapter ‘Roles of Amacrine Cells’). The numbers vary depending on species. The surface layer of the retina contains about 20 types of ganglion cells (see Webvision chapters ‘Morphology and Circuitry of Ganglion Cells’ and ‘Visual Responses of Ganglion Cells’). In primate, impulses from the ganglion cells travel to the brain via more than a million optic nerve fibers, which carry a processed version of images falling on the photoreceptors. The spaces separating the three nuclear layers are the plexiform layers, which differ in anatomy and function. The region containing synapses linking the photoreceptors with bipolar and horizontal cell dendrites is known as the outer plexiform layer (see Webvision chapter ‘Outer Plexiform Layer’); the area where the bipolar and amacrine cells connect to the ganglion cells is the inner plexiform layer (see Webvision chapter ‘Inner Plexiform Layer’).
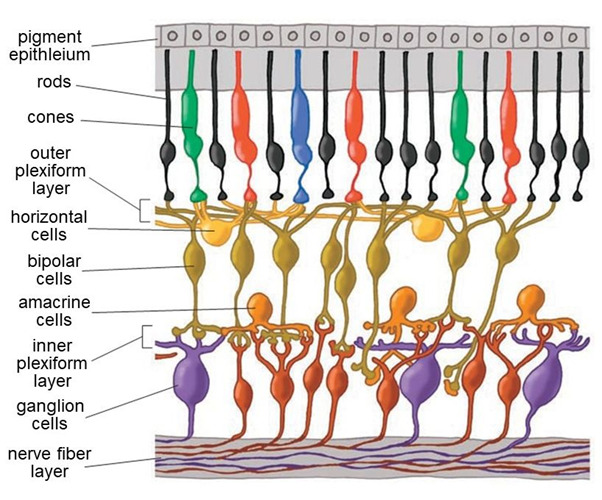
Figure 5. Cells in the retina are arrayed in discrete layers. The photoreceptors are at the top of this rendering, close to the pigment epithelium. The bodies of horizontal cells, bipolar cells and amacrine cells compose the inner nuclear layer. Amacrine cells lie close to ganglion cells near the surface of the retina. Axon-to-dendrite and dendrite to dendrite neural connections make up the outer and inner plexiform layers separating cell bodies layers.
Decades of anatomical studies have shed light on how the retina works. Imaging techniques ranging from Golgi silver staining, first used over a century ago by Santiago Ramón y Cajal (15), to electron microscopy and modern-day antibody staining (16) and selective neurogenetic marking (17) have revealed the shapes, sizes, biochemistries and genetics of the retina’s cell types and show how the different cells may connect to form synapses. Diffusion of permeant tracers have revealed electrical or ‘gap’ junctions between cells (see Webvision chapter ‘Myriad roles for gap junctions in retinal circuits’) and antibody staining techniques identify and localize neurotransmitters, neurotransmitter receptors and transporters. We now know that the neurotransmitter (chemical signal) passed through the vertical pathways of the retina—from photoreceptors to bipolar cells to ganglion cells—is glutamate. The horizontal and amacrine cells send signals using various excitatory and inhibitory amino acids, catecholamines, peptides, nitric oxide, and even hydrogen ions (see Webvision section ‘Neurotransmitters in the Retina’).
Electrophysiological investigations of retinal neurons started almost a century ago. Studies of the frog optic nerve fibers showed that light stimulation produced depolarizing action potentials, like those observed in other neurons (18). However, the first recordings from inside of neurons within the retina by Gunnar Svaetichin in the 1950s showed very odd responses to light (19). Neurons in the outer retina—it was not immediately clear which cells they were—responded to light stimulation not with depolarizing spikes but with slow hyperpolarization. These ‘S potentials’ are now known to originate in the photoreceptors (Figure 6) and are transmitted to the horizontal cells and bipolar cells with which they connect (see Webvision chapter ‘S-Potentials and Horizontal Cells’). The membrane hyperpolarization starts on exposure to light, follows the time course of a light flash, and then returns to the baseline value when the light is off. This reflects the counterintuitive discovery that both rods and cones release the excitatory neurotransmitter glutamate during the dark, when their synapses are depolarized by sodium ions flowing freely across the membranes into photoreceptor outer segments (see Webvision chapter ‘Phototransduction in Rods and Cones’). When exposed to light, membrane cation channels, permeable to positively charged ions like sodium, close, but hyperpolarizing potassium-ion-permeable channels are not gated by light and remain open. The continuing outflow of potassium causes the cells to go into a hyperpolarized state for as long as the light continues to shine on them. This blocks the release of neurotransmitter during light stimulation.
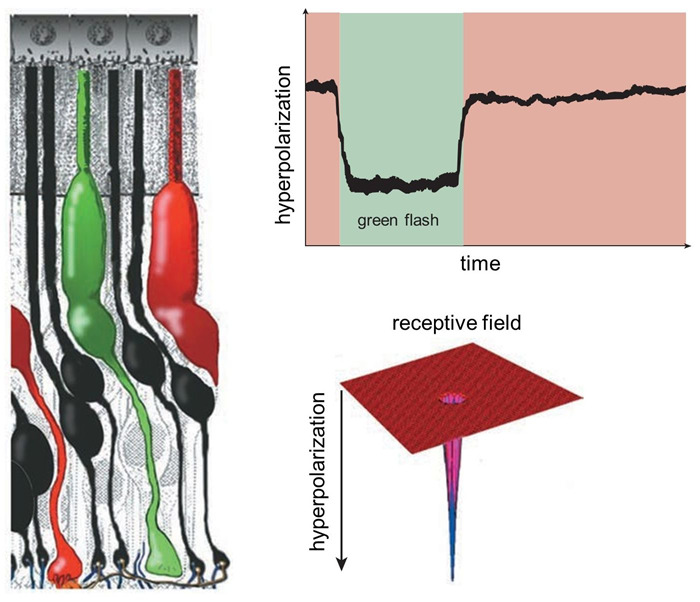
Figure 6. A single green-sensitive cone photoreceptor (left) responds to the presence of green light by becoming hyperpolarized; that is, the membrane’s electrical potential becomes more negative. The hyperpolarization lasts as long as the light flash (top right). The cone only responds to light immediately directed to it, so its receptive field is very narrow (bottom right). These sorts of waveforms in response to light were originally termed ‘S-potentials’.
Although both rods and cones respond to light with a slow hyperpolarizing, ‘S-potential’, response, they report quite different image properties. Rods, detecting dim light, usually respond relatively more slowly, and don’t detect rapid changes in the image. Cones, dealing with bright signals, can detect rapid light fluctuations. In both cases, photoreceptors decompose images into small spatial segments. Both rods and cones mainly respond to light directly over them. Thus, their receptive fields are very narrow (Figure 6), though, as we shall see, these primary light-detecting cells are influenced by other neurons in the retina such as horizontal cells that sense light over a much broader region.
Parallel Processing
Rods and cones selectively encode scenes under dim or bright illumination, respectively. This image information is carried forward to the inner retina by separate rod and cone bipolar cells devoted to each photoreceptor type. This is a specialized form of neural organization called ‘parallel processing’. In the retina local image features, darkness, brightness, direction, quick and slow change are segregated onto separate networks of cells. Parallel processing encodes such image features. Some of this ‘feature detection’ occurs not at the photoreceptors, but at the first synapses of the visual pathway, those between photoreceptors and bipolar cells. Different bipolar cells express different types of receptors for the neurotransmitter glutamate, allowing the cells to respond to photoreceptor input differently (Figure 7) (see Webvision chapter ‘Bipolar Cell Pathways in the Vertebrate Retina’). Some bipolar-cell synapses are tuned to faster and some to slower fluctuations in the visual signal, as some glutamate receptors resensitize rapidly and others more gradually (20). The cells thus prefer either quick or relatively slow changes in the stimulus. These AMPA and kainate receptors respond to glutamate by activating what’s known collectively as the OFF pathway in the visual process, detecting dark features against a lighter background. (Recall that photoreceptors release glutamate in darkness). Other bipolar cells have inhibitory glutamate receptors; in other words, they prevent the bipolar cell from showing excitation when the cell is exposed to the neurotransmitter glutamate. These receptors activate the ON pathway, detecting brighter image features against a darker background, and were shown to be either of a metabotropic type involving G-protein-coupled receptors and biochemical pathways within the bipolar-cell cytoplasm (21, 22) acting on TRPM1 membrane channels (23), or expressing a type of glutamate transporter (EAAT5) which forms an inhibitory chloride channel on binding glutamate (24). The discovery of novel inhibitory glutamate synapses expressed in ON-type retinal bipolar cells led to explanations for the first recognized genetic disease of retinal neural circuits, congenital stationary night blindness (CSNG) (see Webvision chapter ‘Bipolar Cell Pathways in the Vertebrate Retina’).
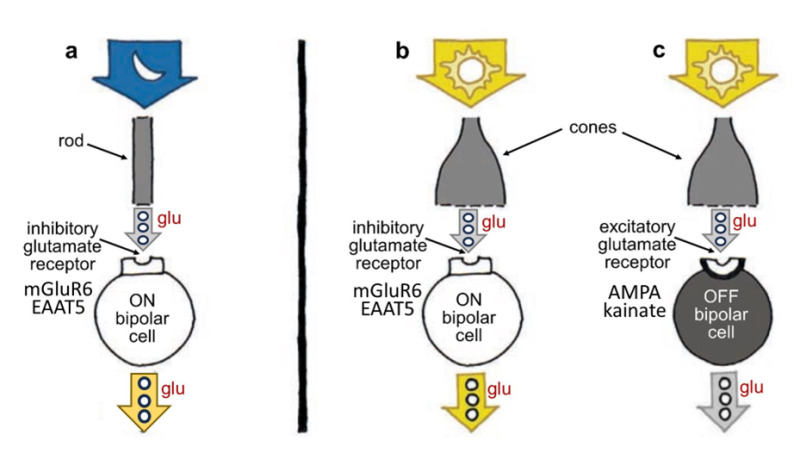
Figure 7. Photoreceptors transmit information to bipolar cells using the molecule glutamate (glu), but different bipolar cells respond differently to the molecule; some are excited and depolarize, whereas others are inhibited and hyperpolarize, depending on the kind of glutamate receptors on their surface. ON bipolar cells, which contact either rods or cones, are excited and depolarized by light (a, b) but inhibited by glutamate; OFF bipolar cells, which in mammals largely only contact cones, are hyperpolarized and inhibited by light (c) but excited by glutamate. Rod and cone bipolar cells all release glutamate from their axon terminals. Contrary to what one might expect, photoreceptors stop releasing glutamate when stimulated by light, in turn causing ON bipolar cell disinhibition leading to release of excitatory glutamate from ON bipolar axons. Release of glutamate from OFF cone bipolar cells is inhibited by light. OFF bipolar cell glutamate receptors are of the ionotropic AMPA or kainate types. ON bipolar cell glutamate receptors are generally of the metabotropic mGluR6 type, though in fish retinas, a glutamate transporter molecule, EAAT5, which forms channels for the inhibitory chloride ion on binding glutamate, is also expressed.
At the retinal output, the ganglion cells, there are parallel sets of visual channels (25) for ON (detecting light areas on dark backgrounds) and OFF (detecting dark areas on light backgrounds). These are qualities of an image that are fundamental to our seeing. For example, we read black letters against a white background using the OFF channels that start in the retina and are excited by the dark letters. Parallel ON and OFF bipolar channels transmit inputs selectively to ganglion cells (see Webvision chapter ‘Bipolar Cell Pathways in the Vertebrate Retina’).
Early in development, the architecture of the inner plexiform layer, full of synapses between bipolar and ganglion cells, shows that synaptic connections become clustered in distinct, layers, with some layers known to selectively process and transmit parallel pathways. Connections occur selectively between ON bipolar cells and ON ganglion cells and selectively between OFF bipolar cells and OFF ganglion cells, with each set of synapses in separate, demarcated strata of the inner plexiform layer (26). Ganglion-cell axons project to the lateral geniculate nucleus of the thalamus, where ON ganglion cells synapse exclusively with ON geniculate neurons, and OFF ganglion cells with OFF geniculate neurons, maintaining the separation of parallel ON and OFF pathways (27). There is a further subdivision within the ON and OFF pathways into at least two well-known types with different response kinetics: the sustained or X type and the transient or Y type ganglion cells (28). These kinetically distinct pathways are also maintained as separate and parallel through selective synaptic contacts within the lateral geniculate nucleus (27).
Sharpening the retinal image
The honing of the image starts at the first synaptic level in the retina, where horizontal cells and bipolar cells process signals from cones. Each horizontal cell receives input from many cones, so its collection area or receptive field is large (Figure 8). Horizontal-cell receptive fields become even broader because their plasma membranes fuse with those of neighboring horizontal cells at gap junctions. The membrane potentials of a whole sheet of cells are intertwined; consequently, horizontal cells respond to light over a very large area (see Webvision chapter ‘S-Potentials and Horizontal Cells). Meanwhile, a single bipolar cell typically receives input from a handful of cones and thus has a medium-size receptive field.
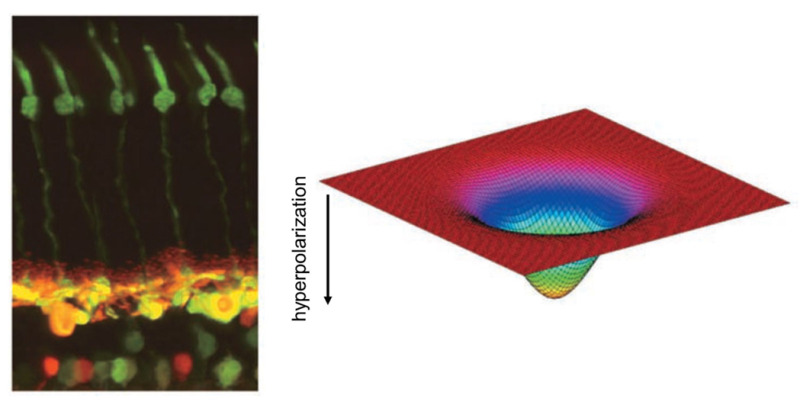
Figure 8. Photomicrograph of a cat retina shows the elongated green-stained cone photoreceptors (left, top) and the horizontal and bipolar cells to which they connect (left, bottom). The horizontal cells are stained yellow; the bipolar and amacrine cells lie below them, stained green and red. Horizontal cells modulate the signals of photoreceptors and bipolar cells. The receptive fields of horizontal cells are smoothly varying and very wide (right) because of electrical coupling among the cells.
Whereas a single cone bipolar cell with its OFF or ON light response would carry a blurry response to its ganglion cell, horizontal cells add an opponent signal that is spatially constrictive, giving the bipolar cell what is known as a center-surround organization (Figure 9). Horizontal cells add an opposing OFF-surround signal to ON center bipolar cells and an opposing ON-surround signal to OFF center bipolar cells. They do this by one of two means. The horizontal cells can either synapse on the bipolar cells directly or feed information back to the cone photoreceptors, which then feed forward surround information to the bipolar cells. The mechanism for feedback from horizontal cells to cones has been debated for decades.
Horizontal-cells are immunoreactive for the inhibitory neurotransmitter GABA (gamma aminobutyric acid), but horizontal-cell modulation of hydrogen ions within the cone synaptic cleft appears to be the major player in the cone feedback pathway (29, 30), and this pH modulation may involve both horizontal-cell GABA receptors (31) and a voltage-sensitive bicarbonate ion channel (17). Hydrogen ions inhibit synaptic L-type calcium channels that control rod and cone transmitter release. This unorthodox circuit from horizontal-cell-to-cone-to-bipolar-cell is still a subject of research and debate in the community of retina scientists (see Webvision chapter ‘S-Potentials and Horizontal Cells’). The mechanism of direct synaptic contact between horizontal and bipolar cells, while not unlikely (32), presents two problems. First horizonal cells express only one neurotransmitter, GABA, but are required to produce inhibitory surrounds for ON bipolar cells but excitatory surrounds for OFF bipolar cells. One explanation is that the chloride channels opened by the horizontal-cell neurotransmitter GABA can be either excitatory or inhibitory, depending on the chloride concentration gradient across the synaptic-membrane GABA receptor (33). The second problem is a lack of typical presynaptic structures such as synaptic vesicles (34) in horizontal cell processes, though oddly, vesicle-associated proteins can be found (35). Recently though conventional synaptic ultrastructure with presynaptic accumulation of vesicles has been found in H1 horizontal cells of avian retinas in contacts with bipolar cells (36).
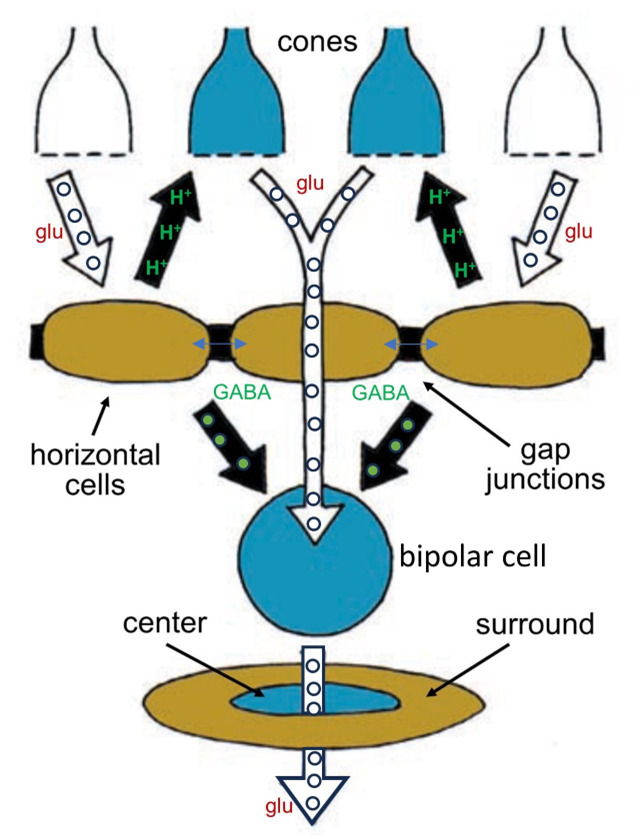
Figure 9. Horizontal cells accumulate information from a wide network of other horizontal cells and influence the signals bipolar cells transmit by adding an opponent surround signal to their receptive fields. The horizontal cells influence bipolar cells both directly, through chemical synapses using GABA, and indirectly, by inhibiting cone synaptic release by altering synaptic pH.
Horizontal-cell function has occupied many vision scientists for decades, and much is now known about the many roles of these cells in the organization of visual messages. The horizontal cells can even make cone and bipolar cell responses color-coded, all apparently through feedback circuits to the cones (37-39). Horizontal cells respond to more than the photoreceptors that link to them. Feedback signals from the inner plexiform layer influence horizontal-cell activity as well. These feedback signals are transmitted via neuromodulators such as dopamine, nitric oxide and retinoic acid (40, 41). The result is that horizontal cells adjust control of the photoreceptor signal under influence of the inner retina. Different lighting conditions allow signaling to become stronger in bright light and weaker in dim light—as well as altering the extent of the horizontal-cell receptive field (42).
At the retinal output, ganglion cells have spatially opponent receptive fields organized as concentric disks (see Webvision chapter ‘Visual Responses of Ganglion Cells’). Much of this spatial profile originates with the bipolar cells, but the amacrine-cell circuitry in the inner plexiform layer conveys additional information to the parallel ganglion cell pathways, possibly sharpening the boundary between center and surround even further than the horizontal-cell input does (43).
In human retinas, two basic types of ganglion cells—ON center and OFF center—form the major outputs of the retina to the visual centers in the brain (Figure 10). ON-center ganglion cells are excited when a spot of light falls in the center of the receptive field and are inhibited when light falls on the field’s periphery. OFF center ganglion cells react in the opposite way: their activity increases when the periphery of their receptive field is bright, and the center is dark but decreases when light falls on the center of the field. (The receptive fields of ganglion cells are modeled spatially as the difference between center and surround Gaussian distributions, giving them a so-called ‘Mexican-hat’ shape (28).)
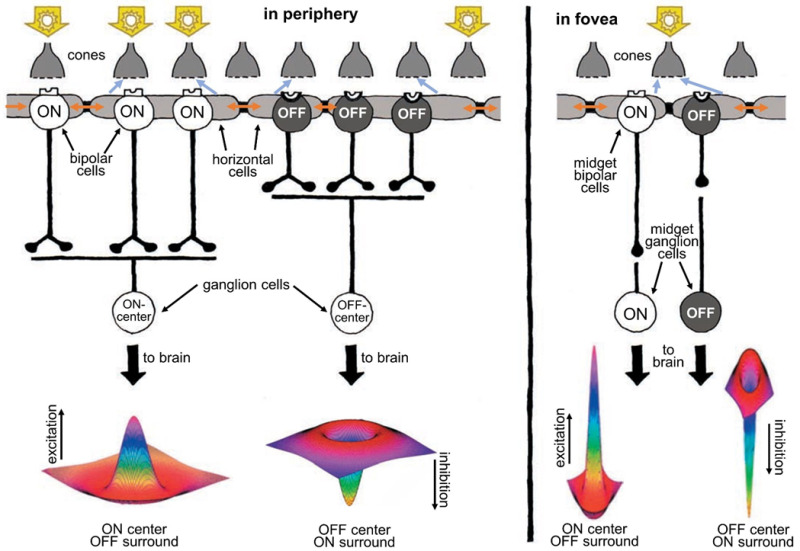
Figure 10. Human retinas, like all vertebrates, have ON-center and OFF-center ganglion cells. ON-center ganglion cells are activated when a spot of light falls in the center of their receptive field, whereas OFF-center ganglion cells fire in response to light falling on their fields’ periphery while leaving the center dark. Horizontal cells, whose syncytial gap junctions (brown arrows) transfer distal signals laterally, convey antagonistic surround signals to cones (blue-gray arrows) and bipolar cells (not shown) through feedback synapses on cones and/or feed forward synapses on bipolar cells and thence onward to ganglion cells. Ganglion cell receptive fields are well represented by a ‘difference of Gaussians’ (Mexican hat) shape, reflecting integration of opposing information from centers and surrounds. This kind of processing helps sharpen the boundaries of images. In the fovea (right), the receptive fields of ganglion cells are much narrower than in the periphery (left). In fact, in the fovea, each ganglion cell carries information from only a single cone.A foveal cone always feeds information to two midget ganglion cells, an ON and an OFF type, each providing signals to the brain. The foveal ganglion cell signal also carries a color message reflecting the spectral type of the individual cone (red or green) that it services.
In contrast to the rest of the retina, the human fovea contains midget ganglion cells (see Webvision chapter ‘Midget pathways of the primate retina underlie resolution and red green color opponency’). The dendritic trees of midget ganglion cells are minute and connected in a one-to-one ratio with individual midget bipolar cells (Figure 10, right). The channel from midget bipolar to midget ganglion cell carries information from a single cone, thus relaying a point-to-point image from the fovea to the brain. Each red- or green-sensitive cone in the central fovea connects to two midget ganglion cells (44), so at the same time each cone can transmit a dark-on-light (OFF) signal or a light-on-dark (ON) message. In this way, the foveal message that goes to the brain carries both spatial contrast and spectral information of the finest resolution.
Short wavelength pathways
Messages from blue-sensitive cones (also called S cones) are not processed in the same way as from red- and green- sensitive cones for some reason, possibly because the blue system is older in evolutionary terms. Blue (or S) cones are found in the retinas of most mammals, cetaceans being an exception (45). The typical mammalian retina complements the blue cone with either a green or red cone, but old-world primates generate both red and green types. Red and green cone opsin molecules, which are also termed L and M opsins respectively, have a common genetic lineage, both grouping with vertebrate long-wavelength opsins. Mutations have separated the spectral sensitivity peaks following gene duplication. Blue-sensitive cone opsins are in a different genetic lineage, which includes the vertebrate UV-sensitive opsins. In primates blue cones transmit information mainly through a single special blue cone bipolar cell pathway (46) to a different type of ganglion cell, which integrates both blue ON and yellow OFF signal pathways as a combined spectral signal (47). But recently, a second parallel ‘blue-OFF’ bipolar pathway has been observed in primate fovea (48).
Extracting other image features
Electrical recordings show that several types of ganglion cells do not have concentric organization. These types are especially prominent in animals whose eyes lack a fovea. This includes most nonmammalian and many mammalian species. Compared with foveate retinas, the nonfoveate species appear to emphasize even more synaptic processing of images in the retina itself before sending a message to the brain (49). Such retinas can immediately synthesize information about image motion and direction of motion (see Webvisionchapter ‘The Anatomy and Physiology of Direction-Selective Retinal Ganglion Cells’). Study of ganglion cells in frog retina led to the concept of ‘feature detection’, that rather than discriminating only bright and dark regions of various dimensions, classes of ganglion cells were tuned to perception of image features more directly relevant to survival and behavior (50). ‘Looming detectors’ were recently added to this list (51). A ‘looming’ image is a ‘threat percept’ requiring avoidance. Ganglion cell classification currently is a mixture of such attempts to project ‘gestalt psychology principles’ onto retinal ganglion cells and the more straightforward center-surround, rod, cone and color representations.
Building Images with Amacrine Cells
There is more to understand about the messages that ganglion cells receive before they transmit a signal to the brain. For that, it is important to appreciate the organization of the inner plexiform layer, where 22 or more different types of amacrine cells make synaptic connections with about 20 different types of ganglion cells (see Webvision chapter ‘Roles of Amacrine Cells’).
It was already clear from Cajal’s description in the 19th century that amacrine-, ganglion- and bipolar-cell dendrites and axons were organized into distinct zones within the inner plexiform layer; Cajal himself divided the inner plexiform layer into five strata (15). But what sorts of synapses were formed among the tangle of intermeshing processes in these strata, and what the structural lamination meant were not immediately apparent. Electron microscopy helped to unravel this neurocircuitry. Now the interconnections of nine types of bipolar cells, 14 types of amacrine cells and eight types of ganglion cells are understood quite well, and the study of such circuits is growing. We can say we are half-way to the goal of understanding the neural interplay between all the nerve cells in the retina.
Much is now known about what types of neurotransmitters different amacrine cells contain and about the organization of receptors at the different synapses. Amacrine cells are largely inhibitory. Amacrine cells are about equally divided between those that use glycine and those that use GABA (gamma-aminobutyric acid) as neurotransmitters (see Webvision section ‘Neurotransmitters in the Retina).
Glycinergic amacrine cells are usually “small field.” Their processes can spread vertically across several strata within the inner plexiform layer, but they extend relatively short distances horizontally. Glycinergic amacrine cells receive information from bipolar cells and transmit information to ganglion cells and to other bipolar and amacrine cells (see Webvision chapter ‘Glycine Receptor Diversity in the Mammalian Retina). Some glycinergic amacrine cells provide interconnections between ON and OFF systems of bipolar and ganglion cells. These are often called ‘cross-over’ cells. The most famous is the AII cell. The AII and a GABA-releasing amacrine cell called A17 are pivotal in the circuitry of rod-based, dim-light vision in the mammalian retina (see Webvision chapter ‘AII Amacrine Cells’). These cells aren’t found in mammalian species that are active solely in daylight and have very few rods—for example, squirrels (52), but there are perhaps distant AII relatives in the retinas of cyprinid fish (53).
In the earlier discussion of ON and OFF pathways emanating from cones, the pathways from rod cells were omitted. Whereas cones connect in a direct pipeline from bipolar cells to ganglion cells, the bipolar cells that receive input from the rods of mammals do not synapse with ganglion cells directly. In mammals, these rod bipolar cells are exclusively rod connected and are all of one type, solely transmitting an ON signal, and synapsing on the AII and A17 amacrine cells, which are intermediaries to get rod signals to ganglion cells. The small-field AII cell collects from about 10-15 rod-connected bipolar cells (54) and transmits a depolarizing message to ON (bright-object-detecting) cone bipolar cells through gap junctions and thence excites the ON ganglion cells with which ON cone bipolar cells connect. This same AII amacrine inhibits both OFF cone bipolar cells and OFF ganglion cells directly (Figure 11). It is as if the AII cells developed in the rod-dominated parts of the retina as an ‘add on’ to existing cone-to-bipolar-cell-to-ganglion cell architecture. Because of appropriate signaling access to both ON and OFF ganglion-cell pathways, optogenetic techniques to develop visual prosthesis are targeting AII amacrine cells (55) (see Webvision section ‘Visual Prosthesis’).
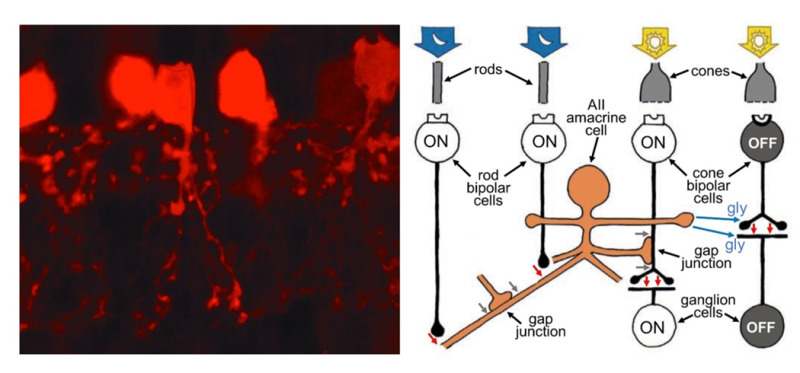
Figure 11. AII amacrine cells play an important role in transmitting information from rod photoreceptors to ganglion cells. As seen with selective staining by parvalbumin antibodies in the hamster retina (left), AII dendrites are bistratified, penetrate the full depth of the inner plexiform layer, but are narrow in lateral spread. AII amacrine cells collect messages from many rod-connected bipolar cells (right), allowing the detection of very dim light. They feed information directly to OFF ganglion cells through inhibitory synapses. They piggy-back onto the ON-cone-bipolar-to-ON-ganglion-cell signal pathway by means of gap junctions with ON cone bipolar cells. In the schematic, red arrows are excitatory glutamatergic synapses between rod bipolar cells and AII amacrine cells and between cone bipolar cells and ganglion cells. Blue arrows are glycinergic (gly) inhibitory synapses from AII amacrine cells to both OFF cone bipolar cells and OFF ganglion cells. Gray arrows are gap junctions allowing signals to pass electrically between AII amacrine cells, and from AII amacrine cells to ON cone bipolar cells (right).
At the same time, thousands of rod-bipolar-cell messages impinge on the A17 amacrine cell (56). It appears that the role of A17 circuits is feedback modulation of transmitter release from rod bipolar axon terminals to AII amacrine cells. This is done on a local microcircuit level, where hundreds to thousands of individual A17 boutons are ‘computational regions’ separately integrating post-synaptic rod-bipolar-cell glutamatergic calcium signals to initiate GABAergic inhibition back onto individual rod-bipolar-cell axon terminals (57, 58). One suspects AII and A17 neurons in the rod pathway are organized to collect and amplify scattered vestiges of light for twilight and night vision.
Wide field amacrine cells sometimes stretch horizontally across the inner plexiform layer for hundreds of microns and interact with hundreds of bipolar cells and many ganglion cells. Such amacrines are usually confined to one of the five different strata of the inner plexiform layer and tile to form elegant meshworks of dendrites. Usually, they are inhibitory, releasing GABA as a neurotransmitter. Sometimes they connect to neighboring amacrine cells by gap junctions, increasing their sphere of influence and the speed at which signals transmit across large areas of retina. Further, their dendrites propagate action potentials to extend the region over which they provide surround inhibition to ganglion cells (43, 59).
Most GABA-releasing amacrine cells also release at least one other neuroactive substance. The secondary substances are usually neuromodulators rather than fast-acting neurotransmitters. The substances include peptides (60)—substance P, somatostatin, vasointestinal peptide and cholecystokinin—as well as the more familiar biomolecules serotonin, dopamine, acetylcholine, adenosine and nitric oxide (61). A variety of receptors have been found on ganglion and bipolar cells—for example, receptors for peptides, nicotine and muscarine (mushroom toxin) in addition to different forms of GABA receptors (see Webvision section ‘GABAC Receptors in the Vertebrate Retina’), indicating that ganglion cells are sensitive to such agents. Most of the neuromodulators are not active at conventional synapses; their release is thought to influence neurons even at a distance by diffusion. Such neuromodulators apparently influence the retinal circuitry under changing light conditions or even cause retinal activity to reflect the different times of day in the circadian clock (62) (see Webvision chapter ‘Melanopsin-expressing, Intrinsically Photosensitive Retinal Ganglion Cells (ipRGCs)’).
A specialized amacrine cell releases dopamine (Figure 12, left) when the retina is stimulated with intermittent flashing light (see Webvision chapter ‘Role of Dopamine in Retinal Function). Dopamine causes the gap junctions among horizontal cells to become uncoupled, reducing the size of their receptive fields (63). Furthermore, the neurotransmitter increases the glutamate receptor conductance on horizontal cells, affecting light responses (64). Again, in the inner plexiform layer, dopamine closes gap junctions, this time the ones that link AII amacrine cells in large networks (65). The resulting uncoupling of the AII cells makes the effective field of influence of the AII rod-system amacrine cells much less significant in brighter conditions (66). Similarly in bright light conditions, another wide-field amacrine cell releases nitric oxide to uncouple the AII cell from the cone-bipolar system (67). Both actions remove the interference of the large-field rod pathway of AII amacrine cells from the narrow-field cone pathways when rod signals are not needed.
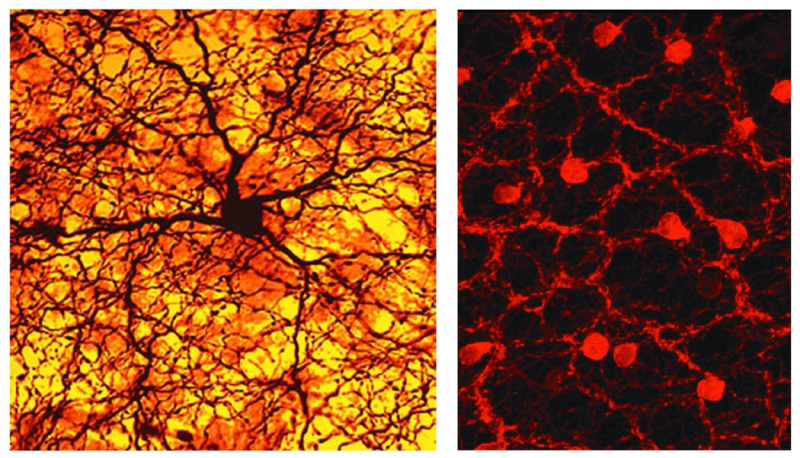
Figure 12. Photomicrographs highlighting two different kinds of amacrine cells show dense networks of dendrites and axons, which send information to various types of cells. One is stained for the global neuromodulator dopamine (left), and the other for the local synaptic neurotransmitter acetylcholine (right).
The ‘starburst’ amacrine releases acetyl choline (Figure 12, right). It is pivotal to the spatio-temporal tuning of directionally selective ganglion cells and has therefore attracted voluminous study (68-71). This amacrine comes in ON and OFF types, each with a ‘starburst’ dendritic branching pattern confined to a characteristic ON or OFF stratum in the inner plexiform layer (72). These strata are so characteristic, they serve as ‘surveyor marker points’ in the inner retina to index the dendritic branching levels of other neurons in the inner plexiform layer.
Summary
The retina provides more than a pixel-by-pixel representation of the visual image projected on it by the eye’s lens. The image is processed and interpreted. Highlights and shadows are distinguished. Wavelength is discriminated. Object boundaries are sharpened, and the direction of motion of objects is calculated. These operations are performed with constancy over ambient light levels that differ by factors of millions utilizing not only rods and cones, but adaptable neural circuits in the inner retina. These functions and more occur within less than half a millimeter thickness of tissue containing three layers of neurons separated by cell free zones containing neural fibers and synaptic contacts.
The above broad sketch of retinal circuitry suggests that the retina is remarkably complex. As vision research advances, we find that the retina itself has an active role in perception. Although we do not fully understand the neural code that the ganglion-cell axons, the retinal output, send as trains of spikes into the brain, we are coming close to understand how ensembles of ganglion cells sense different aspects of the visual scene and how fields of influence on individual ganglion cell types are constructed. Much of the subconscious construction and refinement of the visual images does seem to take place in the retina itself, although the conscious perception of sight is indisputably done in the brain.
Given how much is now known, it might be fair to ask, are we finished with the retina, what has it taught us about neural processing, what was expected, what was unexpected, and are there more surprises on the horizon? Studies of retina have certainly expanded the simple ‘switchboard’ model of neural circuits, unexpectedly increasing the numbers of different varieties of plugs and sockets on the switchboard. This included finding that much of the information transfer in retina is dependent on electrical connections among cells rather than standard chemical synapses (see Webvision chapter ‘Myriad roles for gap junctions in retinal circuits’). For example, the major neural pathway from the rods to ON ganglion cells includes and depends on direct electrical connections. Other fast-acting signals pass directly from amacrine cells into ganglion cells at gap junctions (73). Distant signals pass laterally through horizontal cell gap junctions to modulate cone synapses. Surprisingly also, in retina, the neuron turned out not always to be the functional unit. Functional neural units can be smaller than a single neuron, as in the functionally independent axon terminals of some mammalian horizontal cells (74), or the functionally independent dendritic boutons of A17 amacrine cells (57), which separately integrate local input from bipolar cells. On the other hand, neuromodulators change the milieu of entire neural circuits composed of many cells, acting from a distance by diffusion rather than at closely apposed synapses. These are concepts that need to be considered and integrated with the previous view that all neural interactions take place via neurotransmitters at specialized isolated patches of membrane apposition—that is, synapses. Even some of these ‘conventional’ chemical synapses in retina proved unconventional, with metabotropic rather than ionotropic receptors, and in teleosts, even glutamate transporters (75), serving as postsynaptic glutamate-sensing elements at cone synapses with ON bipolar cells. In another anomalous synapse, the chemical mediating horizontal cell synaptic feedback to rods and cones proved to be none other than hydrogen ions! And there is the continuing suspicion that these same horizontal cells mediate a second type of chemical transmission with bipolar cells utilizing the neurotransmitter GABA, but this communication is done in the absence of synaptic vesicles altogether. In the inner retina, a further recent surprise has been that a previously unknown ganglion cell type appears to function as a giant photoreceptor itself, being ‘intrinsically photosensitive’ without needing input from rods or cones (though receiving such inputs also). This ganglion cell membrane contains light-reactive molecules known as melanopsins, which employ vitamin A, but using a different intracellular phototransduction pathway than rods and cones (see Webvision chapter ‘Melanopsin-expressing, Intrinsically Photosensitive Retinal Ganglion Cells (ipRGCs)). These cells are integral to both pupillary constriction through the olivary pretectal nucleus of the brain and circadian entrainment in the suprachiasmatic nucleus. Given the continued unexpected findings from a close look at retinal circuits, it appears that there may still be much more to learn about how the retina works, and by extension, the brain.
Acknowledgements
This work was supported in part by the Intramural Research Program of the National Institutes of Neurological Disorders and Stroke, NIH.
About the Authors

Helga Kolb, Ph.D. is the originator and chief editor of Webvision. She was born in London and received postgraduate degrees from Bristol University. Helga has been in eye research since 1961, at first doing electrophysiology at the Institute of Ophthalmology, Moorfields Eye Hospital, and then emigrating to the USA. In John Dowling’s lab at the Wilmer Eye Institute, Johns Hopkins University, along with Brian Boycott, she continued work in anatomy of the retina concentrating on understanding the neurons and wiring of the primate retina. Later at NIH in Bethesda, she collaborated with Peter Gouras and Ralph Nelson on joint physiology and electron microscopy of retinal neurons to understand the connections of cell types involved in light, dark and color pathways in cat and monkey retinas. She joined the faculty at the University of Utah Medical School, Salt Lake City in 1979 and is currently a Professor Emeritus in the Department of Ophthalmology and Visual Science at the John Moran Eye Center.
Ralph Nelson Ph.D. (associate editor) was born in Hartford Connecticut. He received a B.A. in Biophysics from Amherst College and a Ph.D. in Biophysics from Johns Hopkins University. He is currently an NIH Scientist Emeritus at The National Institutes of Neurological Disorders and Stroke, National Institutes of Health, Bethesda Maryland. While at Johns Hopkins, he studied ionic mechanisms responsible for light responses of retinal bipolar cells and in his postdoctoral studies at the National Institutes of Health he pioneered the use of intracellular recording for electron-dense staining of retinal neurons in the mammalian retina, a technique that provided insights into the neural circuits for image processing in the inner retina. Helga Kolb and he made major contributions towards the understanding neural pathways that separately process light and dark objects in mammalian retina and the separate neural circuits for nocturnal and diurnal vision. Dr. Nelson currently investigates neural responses in zebrafish retina and the ways in which they are perturbed by genetic mutation.
An American Scientist article on “How the Retina Works” is available here.
References:
1. Kolb, H., How the retina works. American Scientist. 2003.
2. Hubbard, R. and A. Kropf, The action of light on rhodopsin. Proceedings of the National Academy of Sciences. 1958; 44(2):130-139. [PMC free article] [PubMed]
3. Einstein, A., On a heuristic point of view concerning the production and transformation of light. Annalen der Physik. 1905; 17(132):1-16.
4. Jin, M., S. Li, W.N. Moghrabi, H. Sun, and G.H. Travis, Rpe65 is the retinoid isomerase in bovine retinal pigment epithelium. Cell. 2005; 122(3):449-59. [PMC free article] [PubMed]
5. Ahnelt, P.K. and H. Kolb, The Mammalian Photoreceptor Mosaic-adaptive Design. Progress in Retinal and Eye Research. 2000; 19(6):711-777. [PubMed]
6. Peichl, L., Diversity of mammalian photoreceptor properties: adaptations to habitat and lifestyle? The anatomical record. Part A, Discoveries in molecular, cellular, and evolutionary biology. 2005; 287(1):1001-12. [PubMed]
7. Carter‐Dawson, L.D. and M.M. Lavail, Rods and cones in the mouse retina. I. Structural analysis using light and electron microscopy. Journal of Comparative Neurology. 1979; 188(2):245-262. [PubMed]
8. Steinberg, R.H., M. Reid, and P.L. Lacy, The distribution of rods and cones in the retina of the cat (Felis domesticus).Journal of Comparative Neurology. 1973; 148(2):229-48. [PubMed]
9. Famiglietti, E.V. and S.J. Sharpe, Regional topography of rod and immunocytochemically characterized “blue” and “green” cone photoreceptors in rabbit retina. Visual Neuroscience. 1995; 12(6):1151-75. [PubMed]
10. Szel, A., P. Rohlich, A.R. Caffe, B. Juliusson, G. Aguirre, and T. Van Veen, Unique topographic separation of two spectral classes of cones in the mouse retina. Journal of Comparative Neurology. 1992; 325(3):327-42. [PubMed]
11. Easter, S.S., Retinal Growth in Foveated Teleosts: Nasotemporal Asymmetry Keeps the Fovea in Temporal Retina Journal of Neuroscience. 1992; 12(6). [PMC free article] [PubMed]
12. Röll, B., Gecko vision—retinal organization, foveae and implications for binocular vision. Vision Research. 2001; 41:2043-2056. [PubMed]
13. Fite, K.V. and B.C. Lister, Bifoveal Vision in Anolis Lizards. Brain Behavior and Evolution. 1981; 19:144-154. [PubMed]
14. Normann, R.A. and F.S. Werblin, Control of retinal sensitivity I. Light and dark adaptation of vertebrate rods and cones.Journal of General Physiology. 1974; 63(1):37-61. [PMC free article] [PubMed]
15. Cajal, S.R.y., The Structures of the Retina 1972, Translated by S.A. Thorpe and M. Glickstein Springfield: Charles C Thomas.
16. Kalloniatis, M., R.E. Marc, and R.F. Murry, Amino Acid Signatures in the Primate Retina. The Journal of Neuroscience. 1996; 16(21):6807-6829. [PMC free article] [PubMed]
17. Yan, W., M.A. Laboulaye, N.M. Tran, I.E. Whitney, I. Benhar, and J.R. Sanes, Mouse Retinal Cell Atlas: Molecular Identification of over Sixty Amacrine Cell Types. Journal of Neuroscience. 2020; 40(27):5177-5195. [PMC free article] [PubMed]
18. Hartline, H.K., The response of single optic nerve fibers of the vertebrate eye to illumination of the retina. American Journal of Physiology. 1938; 121:400-415.
19. Svaetichin, G. and E.F. MacNichol, Retinal mechanisms for chromatic and achromatic vision. Annals of the New York Academy of Sciences. 1958; 74(1):385-404. [PubMed]
20. DeVries, S.H. and E.A. Schwartz, Kainate receptors mediate synaptic transmission between cones and ‘Off’ bipolar cells in a mammalian retina. Nature. 1999; 397(6715):157-60. [PubMed]
21. Slaughter, M.M. and R.F. Miller, 2-amino-4-phosphonobutyric acid: a new pharmacological tool for retina research.Science. 1981; 211(4478):182-5. [PubMed]
22. Vardi, N., D.F. Matesic, D.R. Manning, P.A. Liebman, and P. Sterling, Identification of a G-protein in depolarizing rod bipolar cells. Visual Neuroscience. 1993; 10(3):473-8. [PubMed]
23. Morgans, C.W., J. Zhang, B.G. Jeffrey, S.M. Nelson, Burke, Neal S, R.M. Duvoisind, and R.L. Brown, TRPM1 is required for the depolarizing light responsein retinal ON-bipolar cells. Proceedings of the National Academy of Science USA. 2009; 106(45):19174-19178. [PMC free article] [PubMed]
24. Grant, G.B. and J.E. Dowling, A glutamate-activated chloride current in cone-driven ON bipolar cells of the white perch retina. Journal of Neuroscience. 1995; 15(5):3852-62. [PMC free article] [PubMed]
25. Shapley, R., Visual sensitivity and parallel retinocortical channels. Annual Review of Psychology. 1990; 41:635-58. [PubMed]
26. Nelson, R., E.V. Famiglietti Jr., and H. Kolb, Intracellular staining reveals different levels of stratification for on- and off-center ganglion cells in the cat retina. Journal of Neurophysiology. 1978; 41(2):472-483. [PubMed]
27. Cleland, B., M. Dubin, and W. Levick, Sustained and transient neurones in the cat’s retina and lateral geniculate nucleus.The Journal of Physiology. 1971; 217(2):473-496. [PMC free article] [PubMed]
28. Enroth-Cugell, C. and J.G. Robson, The contrast sensitivity of retinal ganglion cells of the cat. Journal of Physiology. 1966; 187(3):517-52. [PMC free article] [PubMed]
29. Hirasawa, H. and A. Kaneko, pH changes in the invaginating synaptic cleft mediate feedback from horizontal cells to cone photoreceptors by modulating Ca2+ channels. Journal of General Physiology. 2003; 122(6):657-71. [PMC free article] [PubMed]
30. Wang, T.M., L.C. Holzhausen, and R.H. Kramer, Imaging an optogenetic pH sensor reveals that protons mediate lateral inhibition in the retina. Nature Neuroscience. 2014; 17(2):262-8. [PMC free article] [PubMed]
31. Hirano, A.A., X. Liu, J. Boulter, J. Grove, L. Perez de Sevilla Muller, S. Barnes, and N.C. Brecha, Targeted Deletion of Vesicular GABA Transporter from Retinal Horizontal Cells Eliminates Feedback Modulation of Photoreceptor Calcium Channels.eNeuro. 2016; 3(2). [PMC free article] [PubMed]
32. Behrens, C., S.C. Yadav, M.M. Korympidou, Y. Zhang, S. Haverkamp, S. Irsen, A. Schaedler, X. Lu, Z. Liu, J. Lause, F. St-Pierre, K. Franke, A. Vlasits, K. Dedek, R.G. Smith, T. Euler, P. Berens, and T. Schubert, Retinal horizontal cells use different synaptic sites for global feedforward and local feedback signaling. Current Biology. 2022; 32(3):545-558 e5. [PMC free article] [PubMed]
33. Vardi, N., L.L. Zhang, J.A. Payne, and P. Sterling, Evidence that different cation chloride cotransporters in retinal neurons allow opposite responses to GABA. Journal of Neuroscience. 2000; 20(20):7657-63. [PMC free article] [PubMed]
34. Kolb, H., The organization of the outer plexiform layer in the retina of the cat: electron microscopic observations. Journal of Neurocytology. 1977; 6(2):131-153. [PubMed]
35. Guo, C., A.A. Hirano, S.L. Stella, M. Bitzer, and N.C. Brecha, Guinea pig horizontal cells express GABA, the GABA‐synthesizing enzyme GAD65, and the GABA vesicular transporter. Journal of Comparative Neurology. 2010; 518(10):1647-1669. [PMC free article] [PubMed]
36. Günther, A., V. Balaji, B. Leberecht, J.J. Forst, A.Y. Rotov, T. Woldt, D. Abdulazhanova, H. Mouritsen, and K. Dedek, Morphology and connectivity of retinal horizontal cells in two avian species. Frontiers in Cellular Neuroscience. 2025; 19:1558605. [PMC free article] [PubMed]
37. Packer, O.S., J. Verweij, P.H. Li, J.L. Schnapf, and D.M. Dacey, Blue-yellow opponency in primate S cone photoreceptors.Journal of Neuroscience. 2010; 30(2):568-72. [PMC free article] [PubMed]
38. Connaughton, V.P. and R. Nelson, Spectral responses in zebrafish horizontal cells include a tetraphasic response and a novel UV-dominated triphasic response. Journal of Neurophysiology. 2010; 104(5):2407-22. [PMC free article] [PubMed]
39. Stell, W.K. and D.O. Lightfoot, Color-specific interconnections of cones and horizontal cells in the retina of the goldfish.Journal of Comparative Neurology. 1975; 159(4):473-502. [PubMed]
40. Weiler, R., M. Pottek, S. He, and D.I. Vaney, Modulation of coupling between retinal horizontal cells by retinoic acid and endogenous dopamine. Brain Research Reviews. 2000; 32:121–129. [PubMed]
41. Poitek, M., S. Konrad, and R. Weiler, Effects of Nitric Oxide on the Horizontal Cell Network and Dopamine Release in the Carp Retina. Vision Research. 1997; 37(9):1091–1102. [PubMed]
42. Mangel, S.C. and J.E. Dowling, Responsiveness and Receptive Field Size of Carp Horizontal Cells Are Reduced by Prolonged Darkness and Dopamine. Science. 1985; 229(4718):1107-1109. [PubMed]
43. Protti, D.A., S. Di Marco, J.Y. Huang, C.R. Vonhoff, V. Nguyen, and S.G. Solomon, Inner retinal inhibition shapes the receptive field of retinal ganglion cells in primate. Journal of Physiology. 2014; 592(1):49-65. [PMC free article] [PubMed]
44. Kolb, H., Organization of the outer plexiform layer of the primate retina: electron microscopy of Golgi-impregnated cells.Philosophical Transations of the Royal Society London [Biological Sciences]. 1970; 258:261-283. [PubMed]
45. Peichl, L., G. Behrmann, and R.H. Kroger, For whales and seals the ocean is not blue: a visual pigment loss in marine mammals. European Journal of Neuroscience. 2001; 13(8):1520-8. [PubMed]
46. Mariani, A.P., Bipolar cells in monkey retina selective for the cones likely to be blue-sensitive. Nature. 1984; 308(5955):184-6. [PubMed]
47. Dacey, D.M. and B.B. Lee, The’blue-on’opponent pathway in primate retina originates from a distinct bistratified ganglion cell type. Nature. 1994; 367(6465):731-735. [PubMed]
48. Patterson, S.S., J.A. Kuchenbecker, J.R. Anderson, A.S. Bordt, D.W. Marshak, M. Neitz, and J. Neitz, An S-cone circuit for edge detection in the primate retina. Scientific Reports. 2019; 9(1):11913. [PMC free article] [PubMed]
49. Dubin, M.W., The inner plexiform layer of the vertebrate retina: a quantitative and comparative electron microscopic analysis. Journal of Comparative Neurology. 1970; 140(4):479-505. [PubMed]
50. Lettvin, J.Y., H.R. Maturana, W.S. McCulloch, and W.H. Pitts, What the frog’s eye tells the frog’s brain. Proceedings of the IRE. 1959; 47(11):1940-1951.
51. Temizer, I., J.C. Donovan, H. Baier, and J.L. Semmelhack, A Visual Pathway for Looming-Evoked Escape in Larval Zebrafish. Current Biology. 2015; 25(14):1823-34. [PubMed]
52. Li, W., Ground squirrel – A cool model for a bright vision. Seminars in Cell & Developmental Biology. 2020; 106:127-134. [PMC free article] [PubMed]
53. Hellevik, A.M., P. Mardoum, J. Hahn, Y. Kolsch, F.D. D’Orazi, S.C. Suzuki, L. Godinho, O. Lawrence, F. Rieke, K. Shekhar, J.R. Sanes, H. Baier, T. Baden, R.O. Wong, and T. Yoshimatsu, Ancient origin of the rod bipolar cell pathway in the vertebrate retina. Nature Ecology & Evolution. 2024. [PubMed]
54. Tsukamoto, Y. and N. Omi, Functional allocation of synaptic contacts in microcircuits from rods via rod bipolar to AII amacrine cells in the mouse retina. Journal of Comparative Neurology. 2013; 521(15):3541-55. [PMC free article] [PubMed]
55. Khabou, H., E. Orendorff, F. Trapani, M. Rucli, M. Desrosiers, P. Yger, D. Dalkara, and O. Marre, Optogenetic targeting of AII amacrine cells restores retinal computations performed by the inner retina. Molecular Therapy Methods & Clinical Development. 2023; 31:101107. [PMC free article] [PubMed]
56. Nelson, R. and H. Kolb, A17: a broad-field amacrine cell in the rod system of the cat retina. Journal of Neurophysiology. 1985; 54(3):592-614. [PubMed]
57. Grimes, W.N., J. Zhang, C.W. Graydon, B. Kachar, and J.S. Diamond, Retinal parallel processors: more than 100 independent microcircuits operate within a single interneuron. Neuron. 2010; 65(6):873-85. [PMC free article] [PubMed]
58. Grimes, W.N., J. Zhang, H. Tian, C.W. Graydon, M. Hoon, F. Rieke, and J.S. Diamond, Complex inhibitory microcircuitry regulates retinal signaling near visual threshold. Journal of Neurophysiology. 2015; 114(1):341-53. [PMC free article] [PubMed]
59. Cook, P.B. and J.S. McReynolds, Lateral inhibition in the inner retina is important for spatial tuning of ganglion cells. Nature Neuroscience. 1998; 1(8):714-9. [PubMed]
60. Salio, C., L. Lossi, F. Ferrini, and A. Merighi, Neuropeptides as synaptic transmitters. Cell Tissue Research. 2006; 326(2):583-98. [PubMed]
61. Yazulla, S. and K.M. Studholme, Neurochemical anatomy of the zebrafish retina as determined by immunocytochemistry.Journal of Neurocytology. 2001; 30(7):551-92. [PubMed]
62. Ribelayga, C., Y. Cao, and S.C. Mangel, The circadian clock in the retina controls rod-cone coupling. Neuron. 2008; 59(5):790-801. [PMC free article] [PubMed]
63. McMahon, D.G., Modulation of electrical synaptic transmission in zebrafish retinal horizontal cells. Journal of Neuroscience. 1994; 14(3 Pt 2):1722-34. [PMC free article] [PubMed]
64. Knapp, A.G. and J.E. Dowling, Dopamine enhances excitatory amino acid-gated conductances in cultured retinal horizontal cells. Nature. 1987; 325(6103):437-9. [PubMed]
65. Kothmann, W.W., S.C. Massey, and J. O’Brien, Dopamine-stimulated dephosphorylation of connexin 36 mediates AII amacrine cell uncoupling. Journal of Neuroscience. 2009; 29(47):14903-11. [PMC free article] [PubMed]
66. Bloomfield, S.A., D. Xin, and T. Osborne, Light-induced modulation of coupling between AII amacrine cells in the rabbit retina. Visual neuroscience. 1997; 14(3):565-76. [PubMed]
67. Mills, S.L. and S.C. Massey, Differential properties of two gap junctional pathways made by AII amacrine cells [see comments]. Nature. 1995; 377(6551):734-7. [PubMed]
68. Euler, T., P.B. Detwiler, and W. Denk, Directionally selective calcium signals in dendrites of starburst amacrine cells.Nature. 2002; 418(6900):845-852. [PubMed]
69. Famiglietti, E.V., Synaptic organization of starburst amacrine cells in rabbit retina: analysis of serial thin sections by electron microscopy and graphic reconstruction. Journal of Comparative Neurology. 1991; 309(1):40-70. [PubMed]
70. Briggman, K.L., M. Helmstaedter, and W. Denk, Wiring specificity in the direction-selectivity circuit of the retina. Nature. 2011; 471(7337):183-8. [PubMed]
71. Ding, H., R.G. Smith, A. Poleg-Polsky, J.S. Diamond, and K.L. Briggman, Species-specific wiring for direction selectivity in the mammalian retina. Nature. 2016; 535(7610):105-10. [PMC free article] [PubMed]
72. Tauchi, M. and R. Masland, The shape and arrangement of the cholinergic neurons in the rabbit retina. Proceedings of the Royal Society of London B: Biological Sciences. 1984; 223(1230):101-119. [PubMed]
73. Kenyon, G.T. and D.W. Marshak, Gap junctions with amacrine cells provide a feedback pathway for ganglion cells within the retina. Proceedings of the Royal Society of London. Series B: Biological Sciences. 1998; 265 (1399):919 – 925. [PMC free article] [PubMed]
74. Nelson, R., A. von Lützow, H. Kolb, and P. Gouras, Horizontal cells in cat retina with independent dendritic systems.Science. 1975; 189(4197):137-9. [PubMed]
75. Grant, G.B. and J.E. Dowling, On bipolar cell responses in the teleost retina are generated by two distinct mechanisms.Journal of Neurophysiology. 1996; 76(6):3842-9. [PubMed]